Lipidomic and signaling in platelet production and functions, thrombosis and cell homeostasis (LIPSIPLAT)
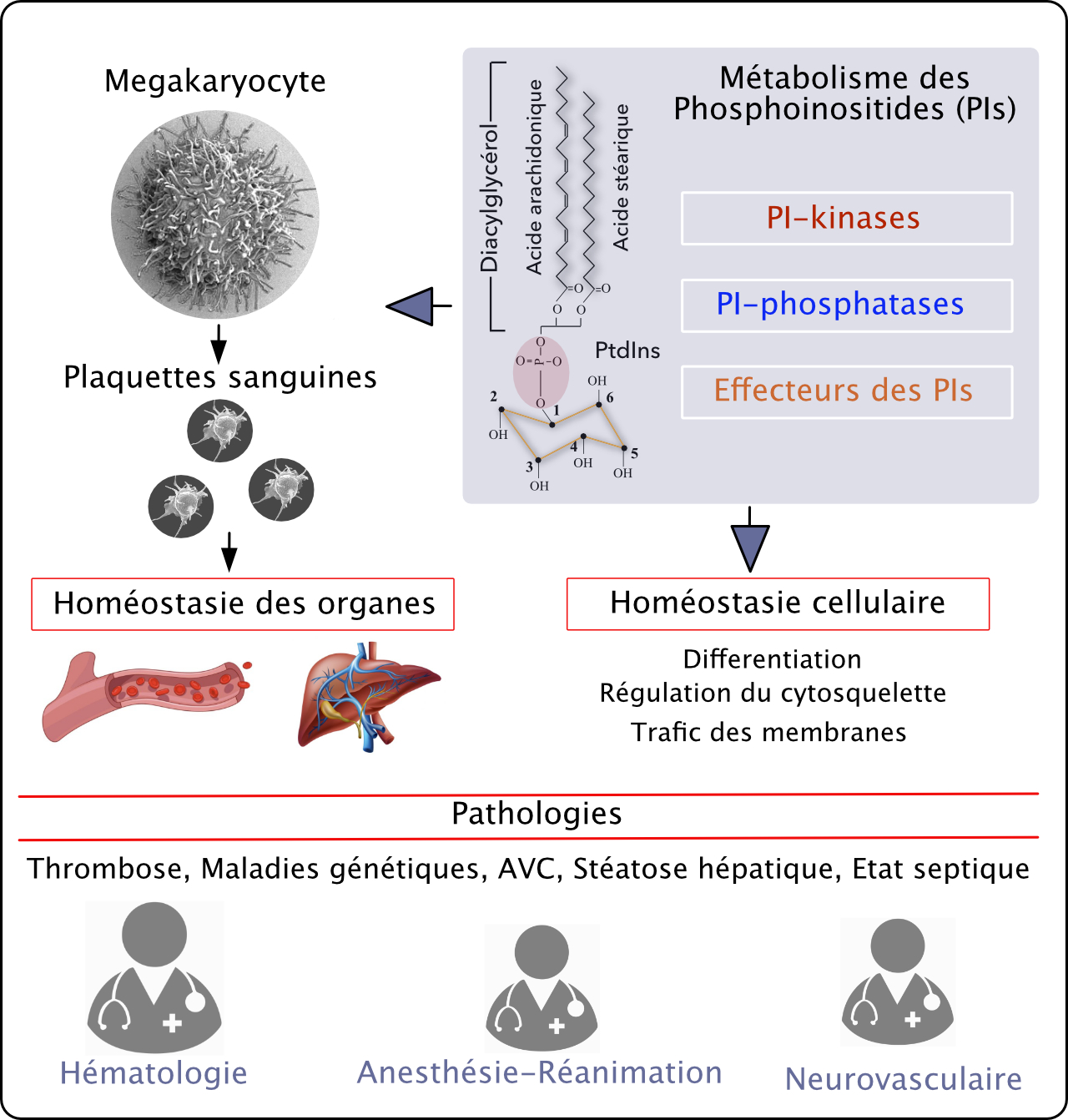
Our laboratory studies phosphoinositide (PI) metabolism in health and diseases. We use integrative and translational approaches to decipher PI signalling and functions in the vascular system (megakaryocytes, platelets and endothelium) and skeletal muscle. We also develop original translational research in the field of cardiovascular diseases in close interaction with clinicians.
TEAM MEMBERS

Bernard PAYRASTRE

Sonia SEVERIN

Marie-Pierre GRATACAP

Julien VIAUD

Christophe COGNARD

Thibault LHERMUSIER

Agnès RIBES

Sophie VOISIN

Fanny VARDON-BOUNES

Gaëtan CHICANNE

Cédric GARCIA

Jean-Marie Xuereb

Anne MAZARS

Laura BOUVET

Oportune KPOTOR

Juline ARDANUY

Christophe FERREIRA

Mathilde MIQUEL

Nadège PREUILH

Claire CONGNET
Phosphoinositide metabolism in cell fate and diseases

Coordinators : Julien VIAUD, Bernard PAYRASTRE
Platelet biology and its integrative function in organ physiology and pathology

Coordinators : Sonia SEVERIN, Marie-Pierre GRATACAP, Bernard PAYRASTRE
Our projects aim to investigate how platelets dialogue with immune cells and with the endothelium to regulate organ functions in normal and pathological contexts. We use cell biology, biochemistry and disease mouse models to investigate the role of platelets in metabolic and cardiovascular diseases and thromboinflammation.
Platelet Signaling in clinical and translational research

Coordinators : Bernard PAYRASTRE, Agnès RIBES, Fanny VARDON-BOUNES, Sophie VOISIN
Our projects aim to establish translational research in close interaction with the National Reference Center of Platelet Pathologies (CRPP) and clinical departments to investigate the molecular basis of acquired and genetic platelet pathologies. We use bench to bedside approaches to implement diagnostic methods, characterize new platelet pathologies and to improve patient care.
Selected publications
PI3KC2β depletion rescues endosomal trafficking defects in Mtm1 knockout skeletal muscle cells.
MTM1-mediated production of phosphatidylinositol 5-phosphate fuels the formation of podosome-like protrusions regulating myoblast fusion.
PIKfyve-Dependent Phosphoinositide Dynamics in Megakaryocyte/Platelet Granule Integrity and Platelet Functions. Caux M, Mansour R, Xuereb JM, Chicanne G, Viaud J, Vauclard A, Boal F, Payrastre B, Tronchère H, Severin S. Arterioscler Thromb Vasc Biol. 2022. Pubmed
Platelet activation and partial desensitization are associated with viral xenophagy in patients with severe COVID-19.
Garcia C, Au Duong J, Poëtte M, Ribes A, Payre B, Mémier V, Sié P, Minville V, Voisin S, Payrastre B, Vardon-Bounes F. Blood Adv. 2022. Pubmed
PI3KC2β inactivation stabilizes VE-cadherin junctions and preserves vascular integrity.
Anquetyl T, Solinhac R, Jaffre A, Chicanne G, Viaud J, Darcourt J, Orset C, Göb E, Kleinschnitz C, Vanhaesebroeck B, Vivien D, Hnia K, Larrue V, Payrastre B, Gratacap MP. EMBO Reports 2021. Pubmed
Adipocyte fatty acid transfer supports megakaryocyte maturation.
Valet C, Batut A, Vauclard A, Dortignac A, Bellio M, Payrastre B, Valet P, Severin S. Cell Reports 2020. Pubmed
Catalytic dysregulation of SHP2 leading to Noonan syndromes impacts on platelet signaling and functions.
Bellio M, Garcia C, Edouard T, Voisin S, Neel B, Cabou C, Valet P, Mori J, Mazharian A, Senis Y, Yart A, Payrastre B, Severin S. Blood 2019. Pubmed
THEY ARE SUPPORTING OUR RESEARCH








Inserm/UPS UMR 1297 - I2MC Institut des Maladies Métaboliques et Cardiovasculaires
1 avenue Jean Poulhès - BP 84225 - 31432 Toulouse Cedex 4
Tél. : 05 61 32 56 00
Horaires
Du lundi au vendredi
8h30 - 12h30 / 13h45 -16h45