CAKUT: Congenital anomalies of the Kidney and the urinary tract
CoordinaTors : Bénédicte Buffin-Meyer, Stéphane Decramer, Jean-Sébastien Saulnier-Blache

Early detection of severe CAKUT
Most forms of CAKUT are detected in-utero by ultrasound. However, although detected at an early stage, in most cases ultrasound does not reliably predict disease progression. Clear-cut information on (post-natal) disease outcome will significantly improve the management of CAKUT pregnancies. We have developed prenatal body fluid (fetal urine and amniotic fluid) peptidome and metabolome analysis that significantly improved on the clinical parameters-based prediction of disease outcome. We are currently validating these urinary peptide signatures in a multicenter European trial called ANTENATAL (clinicaltrials.gov: NCT03116217).
Better understanding of CAKUT physiopathology
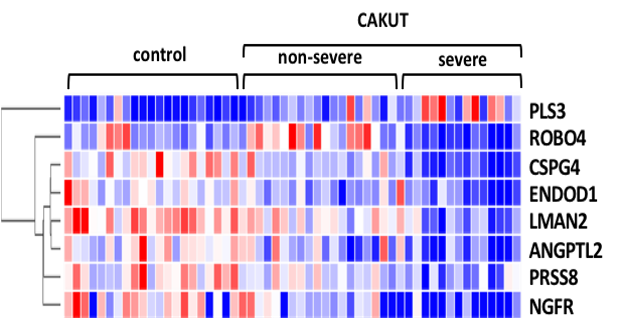
Most CAKUT patients display a wide spectrum of post-natal outcomes ranging from severe, with death in utero, to non-severe with normal kidney function. A better understanding of CAKUT phenotype requires to capture the molecular mechanisms downstream of the genome. We used LC-MS/MS to map the amniotic fluid proteome from control and CAKUT human fetuses, and identified 8 gestational-age independent proteins associated with both the presence and the severity of CAKUT. Among them, p75NTR/NGFR (low affinity p75 neurotrophin receptor) was strongly reduced in abundance with CAKUT severity, while PLS3 (plastin-3) was increased. We demonstrated that invalidation of p75NTR in zebrafish impaired nephrogenesis, while Pls3 knockout mice displayed impairment of glomerular and podocyte, suggesting the possible contribution of p75NTR and PLS3 in CAKUT and its postnatal function consequences.
HNF-1beta
Hepatocyte nuclear factor 1beta (HNF-1ß) anomalies are the most frequently found mutations associated to CAKUT. However, genotype-phenotype correlations in HNF-1ß gene variants have not convincingly been shown and thus the knowledge of the underlying HNF-1ß gene variants does not clearly help the clinical management. We are further exploring this correlation by a large scale survey in the context of ERKnet (www.erknet.org) on individuals with HNF-1ß gene variants and progression of kidney disease.
THEY ARE SUPPORTING OUR RESEARCH










Inserm/UPS UMR 1297 - I2MC Institut des Maladies Métaboliques et Cardiovasculaires
1 avenue Jean Poulhès - BP 84225 - 31432 Toulouse Cedex 4
Tél. : 05 61 32 56 00
Horaires
Du lundi au vendredi
8h30 - 12h30 / 13h45 -16h45